Lucas Sainburg, BE – PhD Student, Biomedical Engineering, Vanderbilt University School of Engineering; VUMC; VUIIS; Liliana Martinez, Undergraduate – Bioengineering – University of Louisville School of Engineering; VUMC; VUIIS; Behnaz Akbarian, BE – Biomedical Engineering – Vanderbilt University School of Engineering; VUMC; VUIIS; Andrew Janson, BE; PhD – VUMC; VUIIS; Dingjie Su, BE – Electrical Engineering – Vanderbilt University School of Engineering; Benoit Dawant, BE; PhD – Biomedical Engineering; Electrical Engineering – Vanderbilt University School of Engineering; Dario Englot, BE; PhD – Biomedical Engineering; Neurology; Radiology – Vanderbilt University School of Engineering; VUMC; VUIIS; Victoria Morgan, BE; PhD – Biomedical Engineering; Radiology – Vanderbilt University School of Engineering; VUMC; VUIIS
Poster Number: 1.457
Rationale:
Epileptic seizures involve widespread network interactions between cortical and subcortical structures where disrupted balance of excitation and inhibition exists.1 Electrical stimulation of the thalamus can reduce seizure frequency in temporal lobe epilepsy (TLE) due to the connectivity between the thalamus and regions within the epileptic network.1 The white matter tracts connecting the thalamus to the rest of the brain can be estimated noninvasively with diffusion-weighted MRI (DWI). Previous studies have shown how large-scale brain networks are altered in TLE.2 This study aims to investigate the effects of TLE on the structural connectivity (SC) of thalamic subregions. We hypothesized that the SC of each thalamic subregion would have distinct patterns of alterations to the rest of the brain.
Methods:
This study included 30 right TLE patients and 105 healthy controls. Subjects underwent a T1-weighted scan (1x1x1 mm3) and DWI (2.5x2.5x2.5 mm3, 92 directions, b = 1600 s/mm2) on a 3T MRI scanner. A total of 115 cortical and subcortical regions of interest and 12 thalamic subregions (6 on each side) were segmented from the T1-weighted scan3.
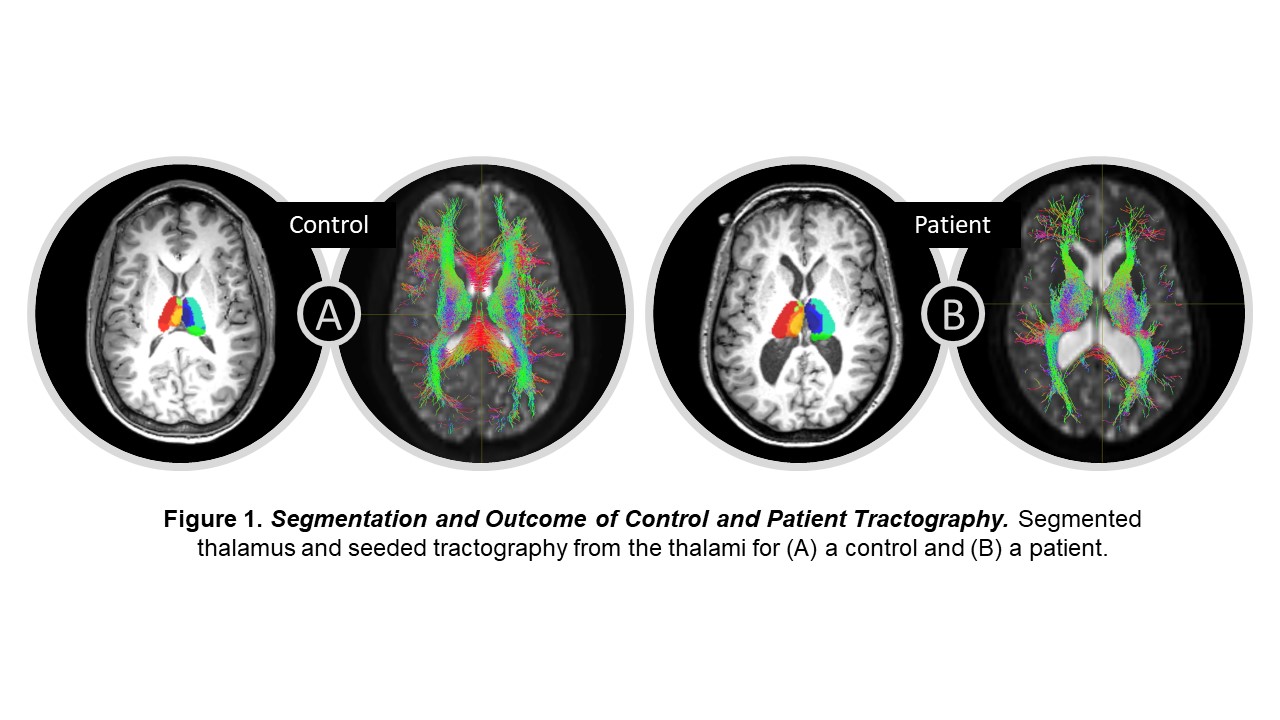
Spherical deconvolution was used to estimate fiber orientation distributions from the DWI data4. The thalamus was then seeded for probabilistic tractography with a minimum streamline length of 20 mm and 2500 streamlines per voxel (Fig 1). SC was computed as the number of streamlines between two regions weighted by streamline length and inverse region size. SC was calculated between each thalamic subregion and all other brain regions to obtain a 12 x 127 structural connectome for each participant.
A two-sample t-test was used to compare the SC, normalized by logarithmic function, between patients and controls at each connection. MATLAB 2021a and MRtrix3 were used for processing and statistics.
Results:
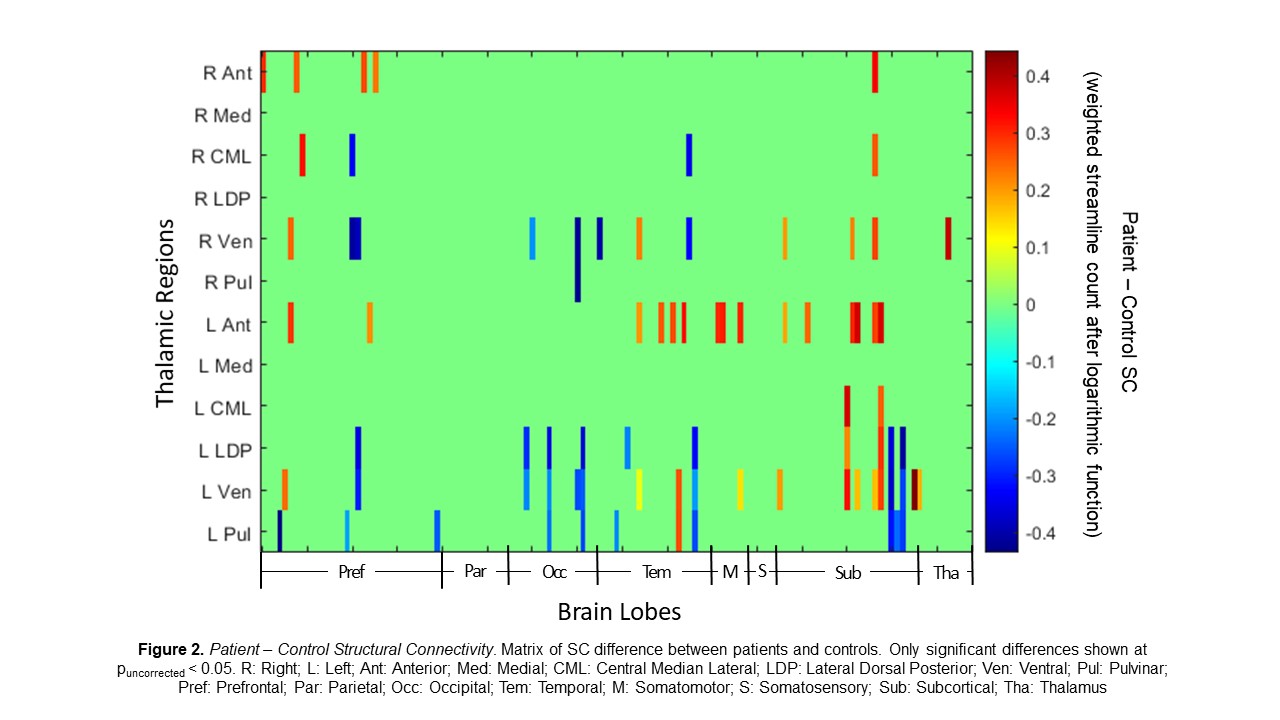
Our results show 79 of the 1524 thalamic connections differed between patients and controls (puncorrected < 0.05; Fig 2). All thalamic subregions, besides medial, had connections that differed between patients and controls; ventral nuclei made up about 43% of the differing connections. The bilateral ventral, lateral dorsal posterior, and pulvinar regions had generally lower SC in patients than in controls while bilateral anterior and central median lateral regions had a generally higher SC in patients than in controls (Fig 2).
Conclusions:
Here we identified the bilateral and widespread aberrant structural connections of the thalamus in TLE which may contribute to seizure generation and spread. These findings may contribute to understanding the thalamus’s role in TLE, potentially leading to improved epilepsy treatment.
Funding:
Funded by NIH T32 EB021937, R01 NS075270, R01 NS108445, R01 NS110130, and R00 NS097618.
References:
1 Norden et al., Epilepsy Behav 2002; 3(3), 219–231.
2 Ronen et al., Magn Res Med 2005; 54 (2), 317–323.
3 Lie Liu et al., Magn Res Imaging 2020; 65, 114-128.
4 Tournier et al., NeuroImage 2019; 202, 116-137.